|
8/15/2023 0 Comments Meaning of resonance in chemistry
Let’s go over those types of resonance in more details. 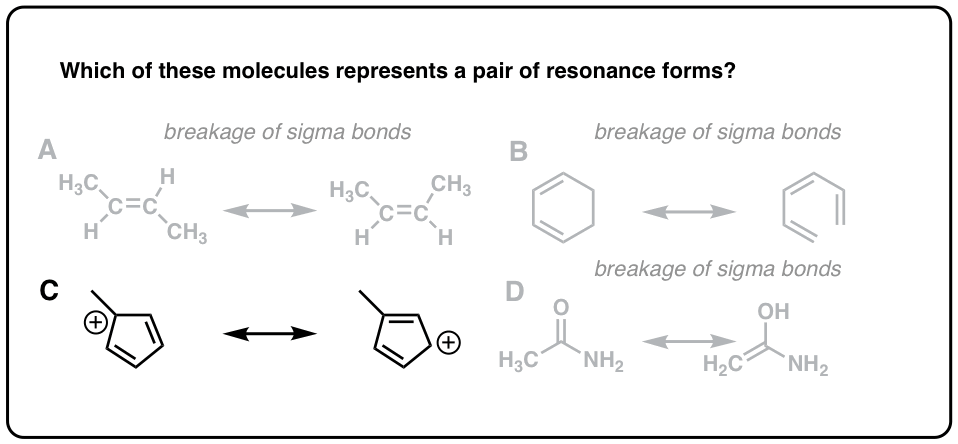
It will explain good 3/4 of reactivity in the course, so it’s an extremely important topic to master. The Resonance stabilization effect (also known as the resonance effect), as briefly mentioned in Section 1. Resonance is one of the most important topic in organic chemistry bonding. While all the examples above are, strictly speaking, conjugated, we’ll only be referring to the multiple π-bonds participating in resonance as “conjugated system” due to their special chemical properties. If there is a negative charge on the lone pair atom, it will transfer that charge to another atom or if there is no charge on the lone pair atom then the. We’ll be talking about those interactions in a lot of details when we discuss the conjugated system. Resonance structures are various forms of the same molecule where the electrons have transferred from one region to another. Two (or more) π-bonds interacting with each other. Resonance is a phenomenon that increases the amplitude or prolongs vibrations in vibrating or oscillating systems. A resonance structure is defined as the energy dependence across the resonance width of the fragment state distributions produced upon resonance decay. However, those are very important for certain types of reactivity, particularly, acid-base chemistry. We’re not going to focus as much on the anions in organic chemistry as on cations. When two different forms of molecules have a similar chemical connection but the distribution of electrons is different. 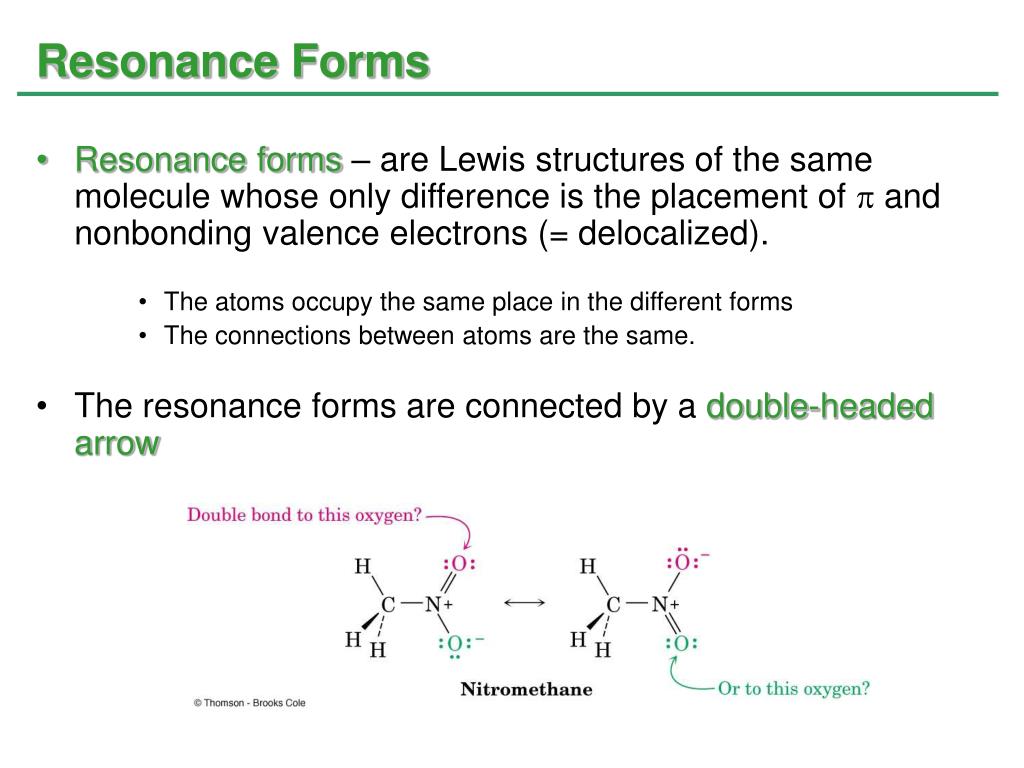
An electron pair and an adjacent π-bond produce allylic or benzylic anions. Those would be examples of allylic and benzylic systems. An interaction between the empty orbital and a π-bond. (iii) The resonating structures must have the same number of paired and unpaired electrons. That generally results in a structure analogous to a simple π-bond. (ii) Resonance occurs only when all the atoms lie in the same plane. An interaction of an empty p-orbital and an adjacent electron pair. There are 4 types of resonance that you’ll have to know within the scope of your organic chemistry course. However, there are situations when three or more orbitals can interact making a much longer and much more complex orbital interaction system. A case of a simple π-bond results from the interaction of the two p-orbitals connecting two atoms. By resonance in organic chemistry we mean an interaction of multiple p-orbitals making a long π-bond spanning multiple atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
